Scandium (Sc) is a high value transition metal that has many industrial applications. While not rare in the earth's crust, Sc-rich minerals are not often found in economically viable concentrations (rarely exceeding a couple hundred ppm), which makes Sc expensive to extract, thus limiting its commercial application. Currently, there are no known economically viable, large-scale primary Sc deposits in the US or Europe. Instead, Sc is produced from wastes that are generated during rare earth, aluminum, titanium, nickel, zinc, tungsten, and uranium refining or in secondary wastes from coal mining and combustion where Sc concentration levels are in the parts per million. For example, coal/coal combustion byproducts (CCP) contain an average Sc concentration of 36-70 ppm along with up to 1000 ppm rare earth elements (REEs). Since Sc is trivalent in nature and behave like some sought-after REEs, there are challenges to extracting valuable Sc from industrial waste streams that are effectively valueless. The forms of waste are usually aqueous and contain Sc plus mixed REEs (lanthanides, Y) as well non-REEs (e.g. Fe, Al, Ca, Mg, Na). There is a great need for a technological change in economics for Sc extraction; perhaps it could be facilitated by the development of novel methods that can efficiently separate Sc from the chemically similar lanthanides and the abundant non-REEs in an environmentally friendly manner.
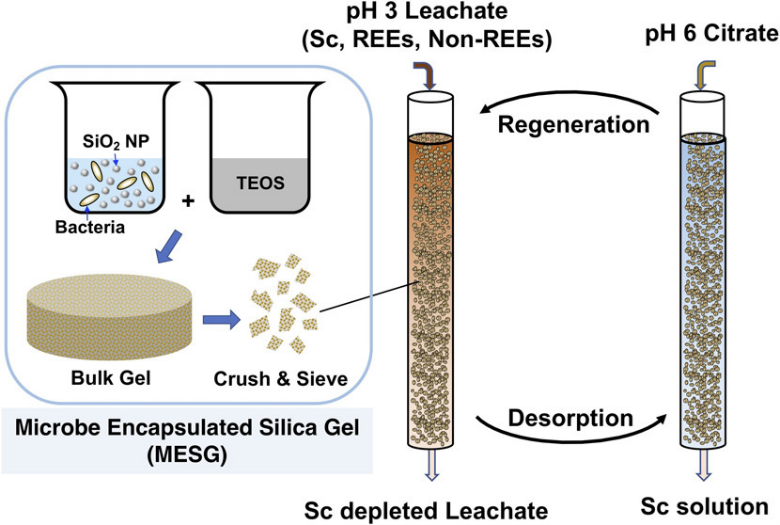
The novel technology developed at LLNL is a new, effective means of separating and concentrating Sc from lanthanides and non-REEs in unconventional, waste-derived feedstocks, thereby transforming an essentially valueless solution into valuable Sc concentrates. The results represent an important advance in the development of an environmentally sustainable alternative to organic solvent-based critical metal recovery approaches. LLNL researchers have developed a biosorption-based approach for the selective recovery of Sc from residue leachates of prepared low-grade industrial feedstocks, including coal/coal byproducts and bauxite residues. Specifically, they developed a cell encapsulation approach whereby Arthrobacter nicotianae (A. nicotianae) bacteria are embedded within a polymer matrix (e.g., Si-sol gel or PEGDA hydrogel) and the resulting microbe particles are loaded into packed-bed columns. A. nicotianae was used due to its exemplary selectivity for Sc relative to REEs and non-REEs at low pH. After this biosorption step which selectively recovers Sc, subsequent REE separation from non-REEs can then be implemented downstream.
Dong Z, Deblonde G, Middleton A, Hu D, Dohnalkova A, Kovarik L, Qafoku O, Shutthanandan V, Jin H, Hsu-Kim H, Theaker N, Jiao Y, Park D. Microbe-Encapsulated Silica Gel Biosorbents for Selective Extraction of Scandium from Coal Byproducts. Environ Sci Technol. 2021 May 4;55(9):6320-6328. (https://doi.org/10.1021/acs.est.0c08632)
- Cost-effective and environmentally sustainable solid liquid extraction (SLE) approach to Sc and REE recovery from dilute solutions.
- One-step separation of Sc from physiochemically similar REEs
- Downstream separation of REEs from non-REEs in subsequent extraction step.
- Extraordinarily effective extraction. This is due to microorganisms’ synthesis and display of high-density surface-accessible functional groups (e.g., carboxylates and phosphates) during growth, facilitating high-capacity Sc adsorption.
- Bio-adsorbed Sc and REEs can be readily recovered using water-soluble environmentally sound organic acids such as citrate or by lowering the pH.
- Biomass can be reused, independent of cell viability.
- Promising results even with complex sample matrices such as leachates from NdFeB hard disk drive magnets, mine tailings, and coal byproducts.
Sc extraction from aqueous waste streams ranging from mine tailings to coal byproducts.
Current stage of technology development: TRL 2
U.S. Patent Application No. 2021/0332392 COMPOSITIONS AND METHODS OF USE THEREOF FOR SCANDIUM SEPARATION FROM RARE EARTH CONTAINING MATERIAL