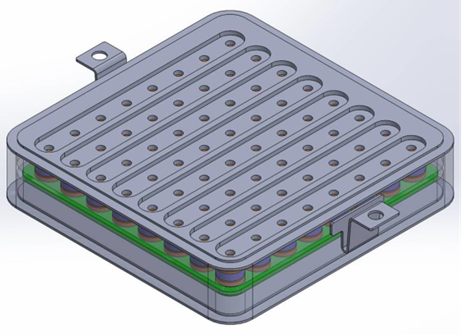
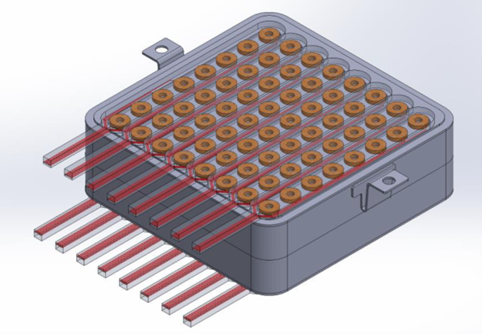
Implantable microelectrode arrays (MEAs) are used in various areas of brain-computer interface (BCI) research. A BCI system is used to interpret electrical signals received from the brain to an external device. One way to record brain signals from the surface of the cortex is through signaling devices that are implanted within the brain. A state-of-the-art BCI system has components that facilitate the signal acquisition and enhancement, provide feature extraction and classification, and a control interface for its operation. A detachable interface for implantable devices is highly desirable.
The current available technology has some major drawbacks:
- Having vertical stacked assemblages limit the number of contacts that can be vertically stacked; conventional connection is limited to 8 to 16 contacts. The unfavorable aspect ratio (~16.7) makes the insertion of contact pins highly challenging since the path a pin must follow is long and narrow (15 mm long and 0.9 mm in diameter). The insertion process is more difficult during surgery where blood and tissue can be hindrances. Frequently, the contact pins break during insertion attempts.
- Pin insertion into the interface is also made more difficult when there are more contacts. The insertion force is linearly dependent on the number of contacts per stack of contact devices. For example, to completely insert a contact pin in a three linearly stacked contact device would require three times the insertion force needed if the device had only a single contact.
- The stacks (of connectors and insulators) must be held in position by external molding. During the molding process, molding compound can seep into electrical contact area, which is very difficult to avoid and correct in the assembly stage. Consequently, an entire system would have to be discarded even if the seepage is isolated to a single, defective component.
Many of the disadvantages of current interface devices can be overcome with LLNL’s novel interface design, which relies on area array distribution where independent interface connector subassemblies are positioned in a planar grid. Not only is the interface device expandable area-wise (without increasing contact force), but it could also be expanded height-wise, with multiple layers of these planar grids of subassemblies. Moreover, within each interface connector subassembly, there are independent pin subassemblies.
When put all together, the arrangement of the subassemblies is the basis for a novel device with a very small form factor yet has numerous contacts, i.e. independent electrical communication channels. The detachable and modular nature of the interface not only provides flexibility in configuration, but also can be a cost-saving measure; if a faulty component is found, only that part is replaced instead of the entire system.
- The high density detachable interface developed at LLNL would allow a significant increase in the number of contacts without tradeoffs such as increased contact pin insertion force.
- Linking the interface device to other system components is less challenging since it can have a smaller aspect ratio for contact pin connections as compared to conventional devices.
- The interface allows module-to-module electrical interface, thus very flexible in terms of configuration to meet the needs of different applications (e.g. aids modular design of implantable sub-systems).
- Additionally, if a distinct part of the system fails, the modular approach allows for replacing of just that defective component rather than having to discard the entire, expensive system.
Biomedical applications involving electrically interfacing to large numbers of microelectrodes implanted in human tissue such as implantable Microelectrode Arrays (MEAs) in Brain Computer Interface (BCI) applications.
Current stage of technology development: TRL-2
- US Patent Application No. 11742605 Apparatus and method for high density detachable electrical interface published 8/29/2023