This is a broad portfolio that includes all aspects of life sciences. Some of the representative areas are bioengineering (brain computer interface, chips to grow and monitor cellular activities, and bioprinting), vaccines and therapeutics (nanolipoprotein particles for the delivery of vaccines and drugs, carbon nanotubes for drug delivery, KRAS inhibitors, and anti-bacterial minerals), medical diagnostics (molecular diagnostics, point-of-care testing, imaging, and forensic), life science instrumentation (PCR instruments, rapid PCR, fluid partitioning, microfluidics, and biosensors), and methods for the extraction and purification of rare earth elements using lanmodulin and other natural/synthetic bacterial proteins.
Portfolio News and Multimedia
In a pioneering project aimed at revolutionizing the design of antibodies and antibody-like molecules through the power of AI, Lawrence Livermore National Laboratory (LLNL) is working to transform medical countermeasure development and biologics discovery. The project is part of an interagency agreement between the Advanced Research Projects Agency for Health and the Department of Energy.
Under the three-year DeNOVO project, LLNL and other institutions (including the University of California, San Francisco, the Institute for Protein Design at the University of Washington, Los Alamos National Laboratory and A-Alpha Bio) will apply high-performance computing and AI to push the boundaries of antibody design.
A new cancer drug candidate developed by Lawrence Livermore National Laboratory (LLNL), BBOT (BridgeBio Oncology Therapeutics) and the Frederick National Laboratory for Cancer Research (FNLCR) has demonstrated the ability to block tumor growth without triggering a common and debilitating side effect.
The discovery of BBO-10203 brings together DOE high-performance computing with AI and biomedical expertise to accelerate drug discovery. LLNL is leveraging its Livermore Computer-Aided Drug Design (LCADD) platform — combining AI and machine learning with physics-based modeling — and world-class DOE supercomputing resources like Ruby and Lassen, to simulate and predict drug behavior long before any compound is synthesized.
Rare-earth elements are essential for many electronic, energy and advanced defense technologies. A research collaboration between LLNL and Pennsylvania State University (Penn State) has generated a portfolio of intellectual property (IP), jointly owned by both organizations, that uses bacterial proteins to pick out critical metal ions.
LLNL has licensed its interest in the joint IP to advanced biochemistry start-up Alta Resource Technologies for commercialization of the resulting technology to transform mineral separation. Similarly, Penn State is negotiating a license agreement with Alta for its interest in the joint IP.
Life Sciences, Biotech, and Healthcare Technologies

Genetic engineering allows insertion and propagation of a foreign genetic element in a heterologous system in a variety of applications such as the creation of genetically modified organisms (GMOs) in the field of agriculture or production of therapeutic proteins and antibodies in the field of healthcare. However, such genetic insertions often lack the required stability to be maintained for…

LLNL researchers have developed an approach to form silicon carbide (and diamond) nanoneedles using plasma etching that create micro pillars followed by chemical etching of the pillars in forming gas containing hydrogen and nitrogen. Combining these two etching processes allow for fabrication of micro- and nanoneedles that are thinner and sharper than conventionally fabricated needles.

After screening at least 50 different cyclodextrins, LLNL researchers found that the drug Subetadex shows enhanced affinities for fentanyl and its analogues. Subetadex is a smaller version of sugammadex and with a smaller central cavity, allows it to bind extremely well to fentanyl by comparison. A pharmacokinetic study showed a rapid clearance of this promising candidate from…

Rather than using genes carried by viruses, LLNL researchers have developed an alternative approach of delivering CAR to T-cells in form of proteins that are carried on the surface of nanolipoprotein (NLP) particles. NLPs are naturally occurring molecules that serve as structural mimics of cell membranes. They can self-assemble and provide a structure or platform for connecting other…
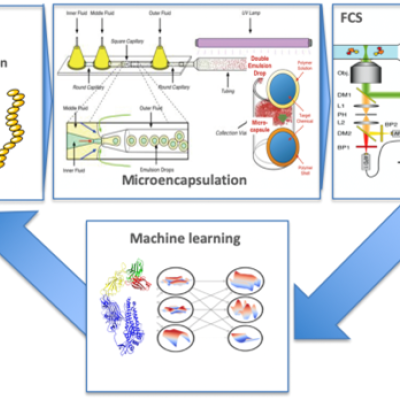
LLNL inventors have developed a method and device for the high throughput screening of various chemical and biological entities using a novel combination of cell-free protein synthesis (in vitro transcription-translation), microencapsulation and optical interrogation (Figure below). This includes automated sample handling, microemulsions, microfluidics manipulation, machine vision,…
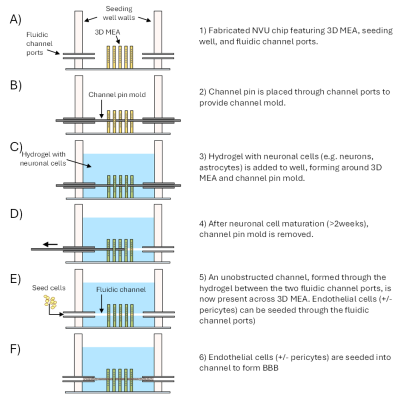
LLNL inventors have developed a proprietary concept and a prototype NVU device that provides an effective in vitro model of the BBB to discover, develop and screen drug candidates for their ability to cross the BBB. The device could also be useful in studying neurological disorders with defects in BBB to understand the mechanism underlying the pathology of the disease that might…

LLNL researchers have designed and developed a novel high-density, high-channel count 3D connector that enables hundreds or thousands of nonpermanent connections within a compact footprint. The connector addresses limitations of currently used conventional approaches that were described previously, which have an artificial ceiling on the number of recording sites of modern devices of no more…
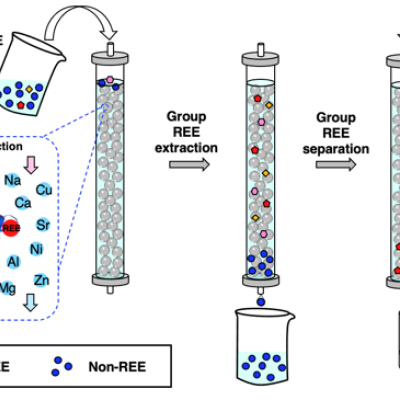
As an important step toward overcoming the technical and environmental limitations of current REE processing methods, the LLNL team has patented and demonstrated a biobased, all-aqueous REE extraction and separation scheme using the REE-selective lanmodulin protein. Lanmodulin can be fixed onto porous support materials using thiol-maleimide chemistry, which can enable tandem REE purification…
LLNL researchers have enhanced and expanded the IDEA technology, which fabricates micron-scale droplets that are then crosslinked in-air (within milliseconds) using a UV light source before collection. An aerodynamically shearing air jet or acoustic vibration force causes rapid droplet formation.

LLNL researchers have discovered that some inexpensive and commercially available molecules used for other applications, could render certain lanthanide and actinide elements highly fluorescent. These molecules are not sold for applications involving the detection of REEs and actinides via fluorescence. They are instead used as additives in cosmetic products and/or in the pharmaceutical…
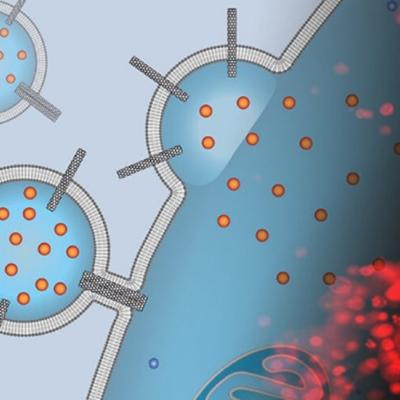
To address shortcomings of current liposome drug delivery systems, the patented innovation uses drug-loaded liposomes containing carbon nanotube porins (CNTPs) inserted into the liposomal membranes for the delivery of the encapsulated drugs. Short CNTPs (10 nm in length) with narrow diameter (0.8 nm) has been demonstrated to facilitate efficient fusion of lipid bilayers resulting in the…

A new approach of developing synthetic antibacterial mineral assemblages can be used as an alternative treatment when traditional antibiotics fail in clinical and agricultural settings. Mineral mixtures can be synthesized with tunable metal release and reactive oxygen species generation that are capable of killing human pathogens and promoting wound healing. One of the key components in the…
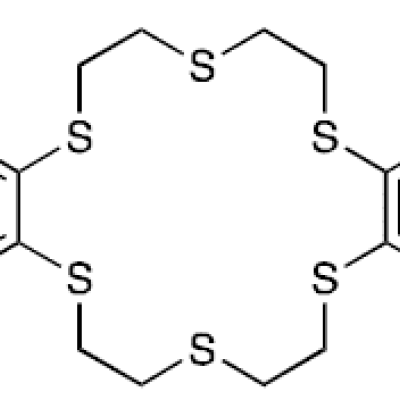
The method described in a pending patent application uses a novel thiacrown (dibenzohexathia-18-crown-6) for efficient extraction of 197m,gHg and 197gHg from irradiated Pt target foils. The separation of 197m,gHg and 197gHg from Pt foils using this novel thiacrown was found to be highly specific. No detectable amount of the Pt foil was seen in the…
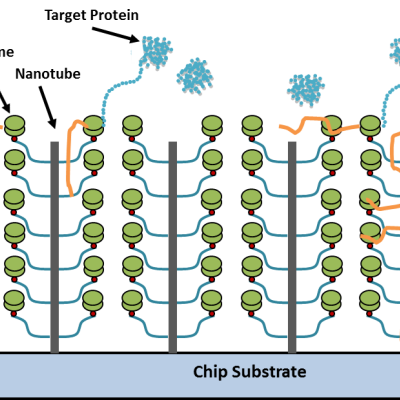
Combining the principles of nanotechnology, cell-free protein synthesis and microfluidics, LLNL researchers have developed a reusable, portable programmable system that can create purified, concentrated protein product in vitro in a microfluidic device containing nucleic acids.
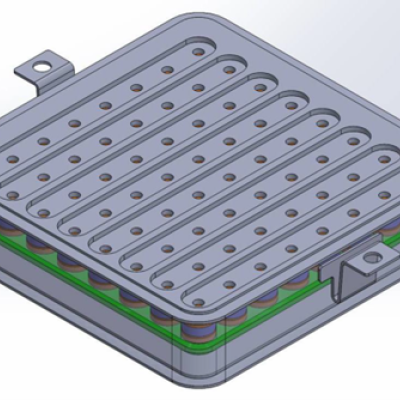
Many of the disadvantages of current interface devices can be overcome with LLNL’s novel interface design, which relies on area array distribution where independent interface connector subassemblies are positioned in a planar grid. Not only is the interface device expandable area-wise (without increasing contact force), but it could also be expanded height-wise, with multiple layers of…

To replicate the physiology and functionality of tissues and organs, LLNL has developed an in vitro device that contains 3D MEAs made from flexible polymeric probes with multiple electrodes along the body of each probe. At the end of each probe body is a specially designed hinge that allows the probe to transition from lying flat to a more upright position when actuated and then…
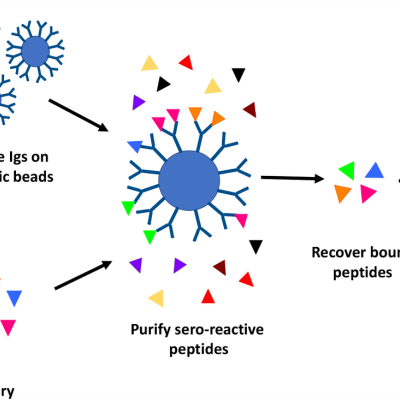
LLNL’s high throughput method involves proteome-wide screening for linear B-cell epitopes using native proteomes isolated from a pathogen of interest and convalescent sera from immunized animals.
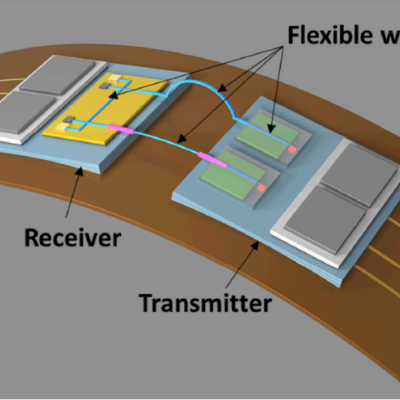
Commercial fiber optic cables are the current standard for carrying optical signals in industries like communications or medical devices. However, the fibers are made of glass, which do not have favorable characteristics for applications that require flexibility and re-routing, e.g. typically brittle, limited selection of materials, dimension constraints.

Solid-state distributed node-based rapid thermal cycler for extremely fast nucleic acid amplification (LLNL Internal Case # IL-12275, US Patent 8,720,209)

Laser heating of aqueous samples on a micro-optical-electro-mechanical system (LLNL Internal Case # IL-11719, US Patents 8,367,976;
LLNL researchers have designed a synthetic, concatemeric bacterial expression vector that expresses a protein sequence that can be digested into a single peptide. The synthetic protein is designed to be secreted outside E. coli cells, and therefore can be purified using a His-tag from the cell supernatant (thereby reducing the need to lyse the cells for purification).

LLNL scientists have created a technology that utilizes electrical means, instead of optical methods, to (1) provide label-free detection of droplet morphology; (2) manipulate droplet position through trapping and actuation; (3) track individual droplets in a heterogeneous droplet population; and (4) generate droplets with target characteristics automatically without optical intercession. The…

This device allows for observation of single cells encapsulated in droplets and provide the ability to recover droplets containing a cell of interest. This system provides the unique capability to monitor droplet contents from a few minutes to hours and overcome the limitations of the fluorescence activated cell sorting (FACS) in the purification of cell populations. The ability of this…
This invention consists of a functionalized membrane (e.g. polyethylene glycol (PEG)) and osmosis or electric potential as a driving force. The PEG membrane provides high biological particles separation and prevents sample for clogging due to the strong hydration of functional polymers layer and their resistance to protein adsorption.

This invention is an improved chromatography device that utilizes the concept of a functionally graded material (FGM) for separation of components. The technology consists of a device that contains a FGM that is patterned to have a gradient in material properties (e.g. chemical affinity, surface chemistry, chirality, pore size, etc.) normal to the direction of flow of the mobile phase. The…

The art described here incorporates a planar integrated optical system that allows for multiple biochemical assays to be run at the same time or nearly the same time. Briefly, each assay can include one or more tags (e.g. dyes, other chemicals, reagents) whose optical characteristics change based on chemical characteristics of the biological sample being tested.

This technology describes a method for partitioning fluid into “packets” between polymeric sheets. The fluid to be partitioned is introduced between two polymeric layers or within a polymeric channel and the layers are sealed together to form an array or sequence of individual milliliter to picoliter samples as shown in figure below. This approach allows a continuous flow of samples through…

LLNL researchers have developed a variation of AMS technology that improves sample preparation, analysis, and cost for AMS. The device involves depositing liquid samples on an indented moving wire and passing the moving wire through a combustion oven to convert the carbon content of samples to carbon dioxide gas in a helium stream. The gas is then directed via a capillary to a high efficiency…

LLNL has invented a new high-throughput assay for sample separation that uses the vibrations of a piezoelectric transducer to produce acoustic radiation forces within microfluidic channels. The system includes a separation channel for conveying a sample fluid containing the different size particles, an acoustic transducer and a recovery fluid stream. The polymeric films containing the…

LLNL researchers have developed an apparatus capable of measuring and recording ultraviolet radiation that uses the Schottky diode/ZnSe/metal type UV sensor. This device can detect both UV-A (320-400nm) and UV-B(280-320nm) radiation. The present invention can also measure and accumulate doses with good sensitivity, and it can also store and make available the readings to be downloaded for…

The invention developed by LLNL researchers proposes to use staged isotachophoresis to improve sample separation. One of the problems with isotachophoresis is that there is a tradeoff between the diameter of the separation column and the ability to isolate a species into a detectable band. For example, wider diameter channels run faster, but narrower channels provide better ability to isolate…
LLNL researchers have devolved a technique to separate or purify samples using electrophoretic separation. This invention corrects the problem associated with pH changes by using the electrode, which contacts the sample, itself a high-conductivity electrolyte made of liquid or gel materials. This will keep the metal surface electrochemistry physically remote from the sample, while…
Researchers at LLNL have developed a more efficient and cost-effective method and system for synthesizing a critical D-aminoluciferin precursor and related compounds. D-aminoluciferin is as active as luciferin and provides a free -NH2 group for functionalization to attach peptide sequences corresponding to the cleavage site of a protease. This allows for the synthesis of bioluminescent probes…

LLNL's technology employs improved sorting strategies related to chip-based droplet sorting. This technology uses electromagnetic fields and non-contact methods to sort and identify monodispersed water-in-oil emulsion droplets in a microfluidic chip-based device. The system selects individual droplets from a continuous stream based on optical or non-optical detection methods as…

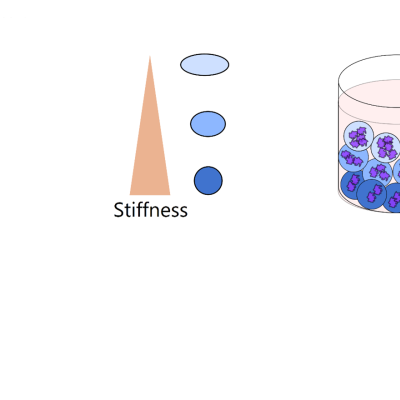
Researchers at LLNL have developed a method to passively sort individual microdroplet samples of uniform size based on stiffness and viscosity. Unlike electrical or optical methods for droplet sorting, this apparatus does not require a measurement step. Instead, particle separation occurs through changes in shearing forces determined by the stiffness of the particles in the microdroplet sample…

LLNL researchers have created a method that uses isotachophoresis for the exclusion and or purification of nucleic acids. Isotachopheresis (ITP) is an electrophoretic separation technique that leverages a heterogeneous buffer system of disparate electrophoretic mobilities. The researchers created a transverse ITP system that offers high-throughput sample preparation as the amount of sample…

The steady-state phenomenon generates thousands of microdroplets per second which is a problem when the stream of droplets needs to be slowed down or stopped. LLNL technology provides a method for generating and trapping microdroplets at a desired location and subsequently stopping the stream of microdroplets without droplet coalescence. These microdroplets can then be chemically reacted,…

The present invention uses magnetic fields to hold particles in place for faster DNA amplification and sequencing. This invention provides a method for faster DNA sequencing by amplification of the genetic material within microreactors, denaturing and de-emulsifying and then sequencing the material while retaining it in the PCR/sequencing zone by a magnetic field. Briefly, nucleic acid…
This invention is designed to sort and identify complex samples using parallel nucleic acid characterization. By isolating single or double stranded nucleic acids derived from complex samples, researchers can sequence previously unknown genetic material to identify novel viruses and organisms. The chip-based microfluidic system achieves this through microdroplet PCR amplification,…
This technology is a photonic detection system developed by researchers at LLNL for the detection of biological or chemical threats with the intention of combining the collection, concentration and detection process onto a single platform. The present invention consists of a porous membrane containing flow-through photonic silicon crystals (see figure).

The described invention is a miniature fluidic device for separating particles suspended within a liquid sample that is introduced into the interior volume of the device. The device uses laminar flow and a combination of gravity and acoustic, electrophoretic, dielectrophoretic, and diffusion-based processes in concert to separate the different particle types and allow them to be collected…
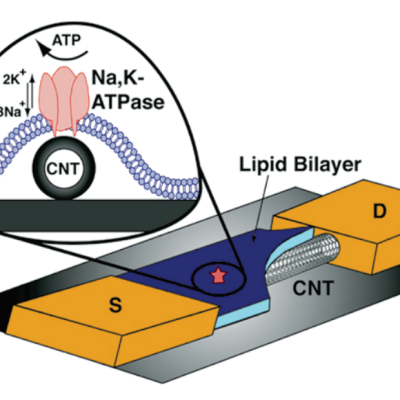
Researchers at LLNL have developed a nanotube sensor (single-walled or multi-walled carbon nanotubes) enclosed within a highly selective lipid bilayer that can detect variations in ion transport using signal amplification generated from the disruption of protein pores across the lipid layer. Changes in the device’s transistor current are recorded by an external circuit with high efficiency as…

Researchers at LLNL have created a new technology for performing pumping and valving operations in microfabricated fluidic systems. Traditional microfabricated devices have some disadvantages that defeat the advantages of miniaturization. For example, they require high power and voltage, and they need specific fluids to work properly and to be broadly applicable. The technology described here…


LLNL has a successful history of developing instruments for detecting and characterizing airborne pathogens. Often, aerosol characterizing instruments require highly focused particle beams with little or no transmission losses. In addition, they need to interface to the sampling environment with a very high sampling rate so that more aerosol particles can be collected and sensitivity can be…

LLNL scientists have designed a rapid PCR technology that incorporates the use of microfluidic thermal heat exchanger systems and is comprised of a porous internal medium, with two outlet channels, two tanks, and one or more exchanger wells for sample receiving. The wells and their corresponding inlet channels are coupled to two tanks that contain fluid with cold and hot temperatures. A…

LLNL has developed a new technology that provides a method for near-instantaneous heating of aqueous samples in microfluidic devices. The technology relates to a heating method that employs microwave energy absorption from a coincident low power Co-planar waveguide or microwave microstrip transmission line embedded in a microfluidic channel to instantaneously heat samples. The method heat…
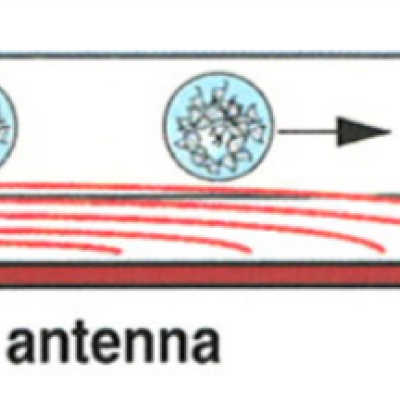
Researchers at LLNL have developed an instantaneous sample heating method to efficiently deposit thermal energy into a continuous stream or segmented microdroplets on a MOEMS device using an optimally low energy, commercially available CO2 laser. The device uses an ideal wavelength (absorption in the far infra-red (FIR) region (λ=10.6 μm)) to instantaneously heat fluidic partitions. The…

Researchers at LLNL have designed a new technology that allows the integration of a large bench-top thermal cycling instrument onto a miniaturized instrument. This instrument is powered and controlled by portable thumb-drive systems such as an USB. USB thumb-drives are commonly used to transfer data from the instrument onto a PC, however, in this new technology the thumb drive becomes the…
LLNL researchers have developed a high-volume, low-cost diagnostic test that is easy to use and provides results in under an hour. The testing platform will provide emergency responders and other medical professionals with the ability to screen individuals using oral and nasal samples, and obtain results in approximately 30 minutes. This point-of-care testing approach will enable rapid triage…

This technology describes a method for performing immediate in-line sample heating to promote the required chemical reactions for amplification, activation, or detection, depending on the thermodynamics of the particular assay involved. The basis of this technology is a method that employ microwave energy absorption to instantaneously heat fluidic partitions without heating the device itself…

LLNL researchers have developed a new method for faster, more accurate, and precise thermal control for DNA amplification. This technology uses sensor-controlled nodes to monitor and cycle materials through a microfluidic heat exchanging system. Thermal energy travels from a power module through thermal electric elements to sample wells. Sensors coupled to each sample well monitor and respond…
LLNL scientists developed novel hydrogels, which are biodegradable soft materials synthesized by a water-soluble polymer. Incorporating silver imparts antimicrobial activity to the material at low concentration compared to currently used silver nanoparticles. Our hydrogels are composed of silver ions instead of silver nanoparticles, which eliminates the toxicity concerns of modern silver…
LLNL researchers have developed a method to quickly and accurately identify the family of a virus infecting a vertebrate via PCR. Universal primer sets consisting of short nucleic acid strands of 7 to 30 base pairs in length were created to amplify target sequences of viral DNA or RNA. These primers can amplify certain identifying sequences of all viral genomes sequenced to date as well as…

LLNL scientists have developed a battery-powered device which is low-cost and multi-chambered for the extraction and amplification of nucleic acids from environmental, clinical, and laboratory samples via loop-mediated isothermal amplification (LAMP). This platform identifies pathogenic bacteria and assists in determining the optimal treatment plan. A multi-chamber amplification cartridge in…

LLNL scientists have developed a high-confidence, real-time multiplexed reverse transcriptase PCR (RT-PCR) rule-out assay for foot and mouth disease virus (FMDV). It utilizes RT-PCR to amplify both DNA and RNA viruses in a single assay to detect FMDV as well as rule out other viruses that cause symptoms in livestock indistinguishable from those caused by FMDV, such as Bovine Herpes Virus-1 (…
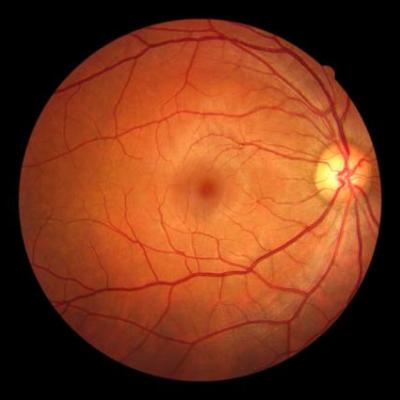
LLNL scientists have developed a method to ensure the accuracy of that tomographic image by applying adaptive optics (AO) to OCT in a single instrument (AO-OCT). AO stabilizes the image being captured by the OCT device by utilizing a Hartmann-Shack wavefront sensor and a deformable mirror, a type of mirror designed to compensate for detected waveform abnormalities (such as ones caused by a…

LNLL scientists have invented a method for multiplexed detection of PCR amplified products which can be completed in a single step. Highly validated species-specific primer sets are used to simultaneously amplify multiple diagnostic regions unique to each individual pathogen. Resolution of the mix of amplified products is achieved by PCR product hybridization to corresponding probe sequences,…

LLNL researchers have developed a portable device which analyzes one or multiple types of body fluids or gases to test for one or more medical conditions. A bodily fluid (such as blood, perspiration, saliva, breath, or urine) is put into a condenser surface and is then separated into both a primarily gas fluid component and a second one that is primarily liquid. These two samples from the same…

LLNL researchers have invented a system for identifying all known and unknown pathogenic or non-pathogenic organisms in a sample. This invention takes a complex sample and generates droplets from it. The droplets consist of sub-nanoliter volume reactors which contain the organism sized particles. A lysis device lyses the organisms and releases the nucleic acids. An amplifier then magnifies the…

This LLNL-developed invention is multiplexed and utilizes the Luminex bead-based liquid array, which contains 100 different unique beads. Oligonucleotide probes with sequences complementary to the target sequences are covalently coupled to these unique beads. These capture beads are mixed with viral samples obtained from the patient via cheek swabbing or a throat wash and subjected to PCR in a…

LLNL has developed a brain-on-a-chip system with a removable cell-seeding funnel to simultaneously localize neurons from various brain regions in an anatomically relevant manner and over specific electrode regions of a MEA. LLNL’s novel, removable cell seeding funnel uses a combination of 3D printing and microfabrication that allows neurons from select brain regions to easily be seeded into…

LLNL has developed a technology that provides near-instantaneous heating of aqueous samples in microfluidic devices. The method heats samples in a focused area within a microfluidic channel on miniaturized chips. The microwave heating device is composed of a waveguide or microstrip transmission line embedded in a microfluidic channel. Aqueous solution microwave heating allows extremely fast…

Lawrence Livermore National Laboratory scientists have developed a signal enhancing microchip apparatus and method that enhances a microfluidic detector's limits by magnetically focusing the target analytes in a zone of optical convergence. In summary, samples are associated with magnetic nanoparticles or magnetic polystyrene coated beads and moved down the flow channels until they are trapped…

The biotech industry aims to move towards an on-chip system for sample generation, amplification and detection of both DNA and RNA based organisms. LLNL has invented a new way of isolating samples in a system.
This invention enables creation of partitioned fluid "packets" between polymeric sheets for chemical separation, DNA amplification or PCR-based DNA detection. The polymeric…
LLNL has developed a novel process of production, isolation, characterization, and functional re-constitution of membrane-associated proteins in a single step. In addition, LLNL has developed a colorimetric assay that indicates production, correct folding, and incorporation of bR into soluble nanolipoprotein particles (NLPs).
LLNL has developed an approach, for formation of NLP/…

Using various excitation wavelengths, a hyperspectral microscope takes advantage of autofluorescence and polarized light scattering from cellular components to obtain composite images that highlight their presence. The light collection efficiency is maximized to achieve image acquisition times and rates suitable for in vivo applications.

LLNL’s BioBriefcase is a compact and portable instrument capable of autonomously detecting the full spectrum of bioagents, including bacteria, viruses and toxins in the air. It uses the state of art technologies to collect, process, and analyze samples to detect, and identify genetic and protein signatures of bioagents.

This technology uses either of two X-ray wave-front sensor techniques, Hartmann sensing and two-dimensional shear interferometry, both of which are capable of measuring the entire two-dimensional electric field, both the amplitude and the phase, with a single measurement. Capturing both the absorption and phase coefficients of the index of refraction can help to reconstruct the image.…




